Comparison of clinical and echocardiographic parameters in patients with hypertrophic cardiomyopathy older and younger than 60 years old
Abstract
Background
Hypertrophic Cardiomyopathy (HCM) is one of the most common genetic myocardial diseases. Transthoracic echocardiography which includes speckle tracking technique is tool for HCM diagnosis and monitoring the course of the disease. The aim of this study was to compare clinical and echocardiographic parameters in HCM patients older and younger than 60 years old (yo).
Materials and methods
We prospectively enrolled 53 HCM patients, who were divided into two groups: younger and older than 60 yo. Clinical parameters, standard echocardiographic indices, as well as strain parameters were assessed and compared between the groups.
Results
The older subgroup was characterized by a higher prevalence of coronary artery disease. In the younger subgroup the incidence of atrial fibrillation was quite high, which occurs far more often than in the general population. Echocardiographic analysis showed worse diastolic function in older, as well as lower volume of the LV. The global longitudinal strain was worse in <60 patients. The 3D strain parameters differed significantly between the groups: the area and radial strains were worse in younger patients.
Conclusions
HCM patients older and younger than 60 yo differ significantly in terms of clinical and echocardiographic parameters.
Citation
Wabich E, Raczak G, Rozwadowska K, Zienciuk-Krajka A, Szymanowicz W, Kozłowski D, Kaleta A, Daniłowicz-Szymanowicz L. Comparison of clinical and echocardiographic parameters in patients with hypertrophic cardiomyopathy older and younger than 60 years old. Eur J Transl Clin Med. 2019;2(1):28-35Introduction
Hypertrophic Cardiomyopathy (HCM) is one of the most frequent genetic diseases, constituting upto 0,2% of general population [1]. In this medical condition the thickness of one or more segments of the left ventricular wall is ≥15 mm and it cannot be explained by the increased left ventricle load [2]. In 60% of adults, the disease is associated with mutations of the cardiac genes coding for sarcomere proteins [2]. Other reasons underlying the development of HCM include congenital metabolic disorders, neuromuscular diseases, mitochondrial diseases, congenital malformations, amyloidosis, drug-induced hypertrophy. However, in 25-30% of patients, the cause of HCM remains unknown. [2]
Transthoracic echocardiography is the most important tool for HCM diagnosis and checking the development of the disease. This method allows to determine the basic parameters describing HCM: hypertrophy of the left ventricular (LV) segments, left atrial (LA) enlargement and LV diastolic dysfunction, which are natural consequences of this disease. Currently available speckle tracking technique additionally allows a detailed assessment of segmental and global LV function [3-7]. This dynamically developing domain of echocardiography allows researchers to assess the function of the LV at very early, preclinical stage of the disease, when the left ventricle ejection fraction (LVEF), assessed by standard echocardiography, is usually preserved [8].
Most of the HCM population studies concern on young patients, before the age of 60. But only few concentrate on elderly patients [9-11], thus it should be emphasized that this is a group in which the HCM diagnosis is more frequent. This situation is most likely a consequence of the growing number of echocardiographic examinations in elderly people and the high availability of echocardiography [11]. Despite the wide availability of echocardiography, there are still no clearly defined guidelines for the management and risk stratification in elderly people with HCM [9-11]. In literature there is also no data concerning to a comparison of older and younger patients with HCM.
Aim
The aim of the present study was to compare clinical and echocardiographic parameters (by standard as well as speckle tracking echocardiography) in HCM persons older and younger than 60 years old.
Material and methods
Fifty three HCM patients were prospectively enrolled [2]. Exclusion criteria were: <18 years of age, with NYHA class IV heart failure, coronary or hemodynamic instability at the moment of enrollment, a history of hospitalization due to cardiological reasons within the 3 months prior to enrollment and poor clinical prognosis.
Echocardiography
Echocardiography was performed in every person (GE VIVID E9; probe M5S; offline analysis using Echo-PAC). During the same examination, 3D datasets were obtained in apical view using a matrix-array 3D transducer (4V). Standard echocardiographic parameters were measured according to relevant guidelines [12-14].
Standard echocardiographic parameters
The LA anterior-posterior size (LADs), LV end-diastolic diameters (LVEDD), LV end-systolic diameter (LVESD), intraventricular septal (IVS) and posterior wall (PW) were measured in parasternal view. In sum, relative wall thickness (RWT) = (IVS + PW) / LVEDD. Whereas in apical four-chamber view the right ventricular internal diameter (RVID), tricuspid annular plane systolic excursion (TAPSE) by M-MODE technique, and LV volumes were measured. LVEF was calculated using the biplane Simpson’s method. The mitral diastolic velocity (E and A velocities, E deceleration time – DT), Em (the average from septal and lateral by tissue Doppler), and E/Em ratio were calculated. Tissue doppler was used for measurement the maximum systolic annular velocity (S') for RV (S’RV).
Speckle tracking parameters
Two-D longitudinal speckle tracking analysis was performed with the use of three endocardial markers (at the end-diastolic frame: at apical four- and three-chamber views). EchoPAC software automatically tracked the contour of endocardium and later this was verified (and corrected if necessary) as described in literature [15]. To quantify the apical and basal LV rotations, appropriate short-axis planes were scanned [16]. Counter-clockwise rotations (from the LV apex) was calculated as a positive; whereas clockwise rotations - a negative value. LV twist was the highest net difference in degrees between the apical and basal rotation. LV torsion was defined as LV twist indexed by LV diastolic longitudinal length. Peak systolic (peak rotation) velocity and early diastolic apical and basal rotation (untwisting) velocity were derived from rotation rate curves.
LA strain 2D
Two- and four-chamber apical views were determined by finding the largest long-axis. Due to the absence of an LA-specific software, we used software dedicated to LV analysis as described in the literature [15, 17].
3D parameters
The appropriate software (4DQ analyses) was used for LV end-systolic, end-diastolic volumes, LVEF and 3D strains measurement. Calculation of 3D area strain (3D-AS) which means global longitudinal plus circumferential strain, and 3D global radial strain (3D-RS) was performed by averaging the end-systolic segmental values when at least 14 segments had acceptable tracking [16, 18].
We divided all patients in 2 groups according their age: below 60 years of age (<60) and 60 years or older (≥60). The further analysis were compared between these subgroups. The protocol of the study was accepted by the Local Ethics Committee and written informed consent was obtained from all participants.
Statistical analysis
All continuous data were presented as median (25th-75th), categorical data in proportion. Mann-Whitney U test and Pearson’s chi-square test were used to compare the differences between the groups. P value <0.05 was considered statistically significant. The statistical analysis was performed using the R 2.15.2 environment.
Results
From enrolled 53 patients, subgroups <60 yo and ≥60 yo was formed of 39 and 14 patients respectively. The higher incidence of hypertension, type 2 diabetes and hyperlipidemia was observed in the older group, differences in frequency of ischemic heart disease reached statistically significant values (Table 1). Particularly noteworthy is the frequency of atrial fibrillation, which was similar in both subgroups, older and younger. However, the 5-year risk of sudden cardiac death, calculated using the European Society of Cardiology calculator was lower in older people [2]. This result was of borderline statistical significance.
Table 1. Clinical characteristics of < 60 and ≥ 60 HCM patients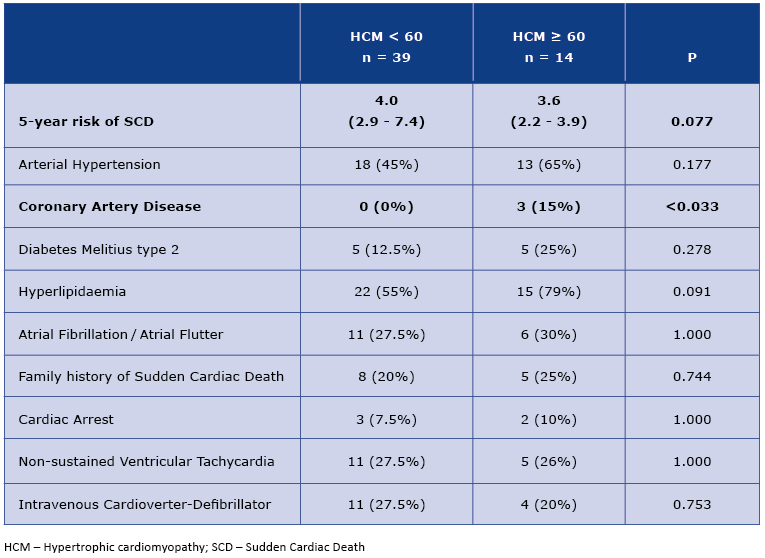
Analysis of the standard echocardiohraphic parameters showed a worse diastolic function in the older subgroup (values of E/A, decT, Em, E/Em, A pulm and A dur pulm parameters - see Table 2). The 2D-peak LV untwisting rate parameter which corresponds to the diastolic function was worse in the older group, although the differences were not reach statistical significance (Table 3). In ≥60 HCM patients we found significantly lower LV volumes (Table 2).
Table 2. Standard echocardiographic parameters in < 60 and ≥ 60 HCM patients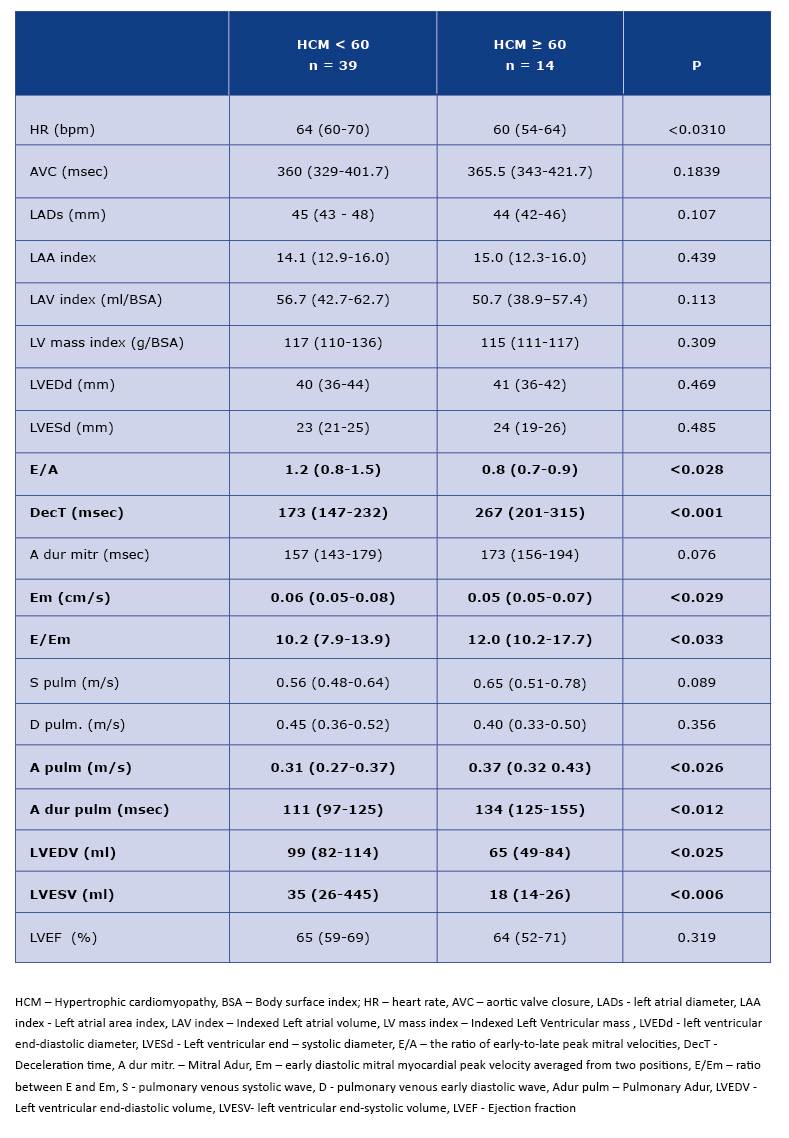
The 2D speckle tracking analysis did not reveal statistically significant differences between the subgroups, although it should be noticed, that the longitudinal strain, measured by 2D-GLS, was worse in the subgroup <60. The 3D strain parameters were significantly worse in younger HCM patients.
Table 3. 2D- and 3D – strain parameters in < 60 and ≥ 60 HCM patients

Discussion
Our findings revealed a number of clinical and echocardiographic differences among patients with HCM. As expected, our older patients (≥60) had more comorbidities (particularly coronary artery disease), which was in accordance with the data from the literature and consistent with population data [20]. The incidence of atrial fibrillation (AF) among the study population deserves the special attention (Table 1). In the general population the frequency of AF increases with age [21], whereas in our study every 3rd-4th patient in both age groups had history of AF. This arrhythmia is the most common complication of HCM, thus rigorous monitoring for its occurrence is necessary and the anticoagulant treatment should be administrated regardless the CHA2DS2-VASc score [2, 21].
The early appearance of diastolic dysfunction in HCM patients is a common symptom described in the literature [19, 22]. In the healthy population diastolic dysfunction progresses with age and is a proof of the natural aging process in the myocardium [23]. As a matter of fact, in our study the diastolic dysfunction was more expressed in patients ≥60, but also in younger patients those parameters were worse than in a healthy population of corresponding age [19, 22-23]. It is known that the parameters of size and function of LA deteriorate with the progression of diastolic dysfunction. It is also known that the processes of fibrosis and hypertrophy of LV always goes straightforwardly with remodelling of LA [24]. This statement has a proven predictive value; for example Iioi et al demonstrated that serious cardiac events in HCM patients were independently related with increased LAVI [4]. The results of our study confirm that dependencies: our patients in both age groups had increased LADs, LAAI and LAVI in comparison to the healthy population from the literature [4].
An important parameter evaluated in our study is LA speckle tracking analysis. The data from literature indicate the potential usefulness of the speckle tracking technique in predicting the cardiac events among HCM patients. For example Fujimoto et al proved that LA peak longitudinal strain <20.3% is associated with an increased risk of heart failure and atrial fibrillation in the follow up [17, 25]. A similar observation was also presented by the other authors [4, 10, 15]. In our study, in both age subgroups the LA peak longitudinal strain was <20.3% (Table 2), which may have significant clinical implications and requires further research.
LVEF in persons with HCM is usually preserved or increased when compared with the healthy population [26]. Our study confirms that the median value of LVEF in both age subgroups was >50% (the lower limit of normal range) (Table 2). However, LV longitudinal strain is usually worse in HCM patients than in healthy people which is in agreement with our results. The median value of 2D-GLS for an older and younger subgroups stood at -16.0% and -14.8% respectively, whilst in healthy population the value of 2D-GLS stands at -20% and more [27]. Debonnaire et al present interesting analyses, which show that the values of 2D-GLS <14% and LAVI >34ml/m2 are independent predictors of adequate therapy of cardiac implantable cardioverter- defibrillators [3, 6]. In turn, Verge et al specifies the strain of the myocardial areas affected by hypertrophy hypertrophy as a predictor of sudden cardiac death [6].
Currently we can observe rising interest in myocardial strain analysis with the use of 3D echocardiography [18]. Our study presents interesting results in that regard: the area strain parameters (obtained by addition of longitudinal and circumferential strains), as well as the radial strain, were statistically significantly worse in the subgroup <60 yo (Table 3), which is in agreement with the literature and directly indicates on worse systolic function in younger subgroup [18].
Other interesting parameters, measured by the speckle tracking technique, are LV twist and torsion, which according to Zhang et al may be used as an echocardiographic marker of fibrosis grade in HCM patients [5, 7]. The above-mentioned study shows that the LV twist and torsion are much higher in patients with myocardial fibrosis confirmed by the cardiac magnetic resonance examination. The authors also suggest that thanks to these parameters, the speckle tracking echocardiography may be useful for assessing the grade of myocardial fibrosis [7]. In our study there were no differences between the age subgroups in terms of the extent of LV twist and torsion, although this topic undoubtedly requires further research.
Our study has a low sample size, which is partially the result of a highly selective enrollment process to achieve the best quality of images (FR from 50 to 80 frames/sec). Thus, larger study groups are strongly needed to verify our results. Secondly, our results were not verified by another imaging technique, for example cardiac magnetic resonance.
Conclusions
Older HCM patients are characterized by a higher incidence of coronary artery disease, while younger ones by atrial fibrillation, which occurs far more often than in the general population. An echocardiographic analysis indicates worse diastolic function in group ≥60 yo and worse systolic function (measured by speckle tracking technique) in group < 60 yo. The clinical relevance of analysed echocardiographic parameters, especially in terms of prognosis of the HCM patients, requires further research with a larger sample size.
References
| 1. |
Gersh BJ, Maron BJ, Bonow RO, Dearani JA, Fifer MA, Link MS, et al. 2011 ACCF/AHA Guideline for the Diagnosis and Treatment of Hypertrophic Cardiomyopathy. J Am Coll Cardiol. 2011;58(25):e212-e260.
|
| 2. |
Elliott PM, Anastasakis A, Borger MA, Borggrefe M, Cecchi F, Charron P, et al. 2014 ESC Guidelines on diagnosis and management of hypertrophic cardiomyopathy. Eur Heart J. 2014;35(39):2733–79.3.
|
| 3. |
Debonnaire P, Thijssen J, Leong DP, Joyce E, Katsanos S, Hoogslag GE, et al. Global longitudinal strain and left atrial volume index improve prediction of appropriate implantable cardioverter defibrillator therapy in hypertrophic cardiomyopathy patients. Int J Cardiovasc Imaging. 2014;30(3):549–58.
|
| 4. |
Iio C, Inoue K, Nishimura K, Fujii A, Nagai T, Suzuki J, et al. Characteristics of Left Atrial Deformation Parameters and Their Prognostic Impact in Pa- tients with Pathological Left Ventricular Hypertrophy: Analysis by Speckle Tracking Echocardiography. Echocardiography. 2015; 8;32(12):1821–30.
|
| 5. |
Popović ZB, Kwon DH, Mishra M, Buakhamsri A, Greenberg NL, Thamilarasan M, et al. Association Between Regional Ventricular Function and Myocardial Fibrosis in Hypertrophic Cardiomyopathy Assessed by Speckle Tracking Echocardiography and Delayed Hyperenhancement Magnet- ic Resonance Imaging. J Am Soc Echocardiogr. 2008;21(12):1299–305.
|
| 6. |
Vergé M-P, Cochet H, Reynaud A, Morlon L, Peyrou J, Vincent C, et al. Characterization of hypertrophic cardiomyopathy according to global, regional, and multi-layer longitudinal strain analysis, and prediction of sudden cardiac death. Int J Cardiovasc Imaging. 2018;34(7):1091–8.
|
| 7. |
Zhang H-J, Wang H, Sun T, Lu M-J, Xu N, Wu W-C, et al. Assessment of left ventricular twist mechanics by speckle tracking echocardiogra- phy reveals association between LV twist and myocardial fibrosis in patients with hypertrophic cardiomyopathy. Int J Cardiovasc Imaging. 2014;30(8):1539–48.
|
| 8. |
Fijałkowski M, Nowak R. Speckle tracking technique; Clinical Echocardiography; Lipiec P, Hoffman P et al; 1 ed., Warsaw, Total Project, 2017:50-57.
|
| 9. |
Maron BJ, Casey SA, Haas TS, Kitner CL, Garberich RF, Lesser JR. Hypertrophic Cardiomyopathy With Longevity to 90 Years or Older. Am J Cardiol. 2012;109(9):1341–7.
|
| 10. |
Maron BJ, Casey SA, Hauser RG, Aeppli DM. Clinical course of hypertrophic cardiomyopathy with survival to advanced age. J Am Coll Cardiol. 2011; 58:2313-2321.
|
| 11. |
Maron BJ, Rowin EJ, Casey SA, Haas TS, Chan RHM, Udelson JE, et al. Risk Stratification and Outcome of Patients With Hypertrophic Cardiomyo- pathy >=60 Years of Age. Circulation. 2012;127(5):585–93.
|
| 12. |
Belghiti H, et al. Automated function imaging: a new operator-independent strain method for assessing left ventricular function. Arch Cardio- vasc Des. 2008; 101(3): 163-169.
|
| 13. |
Leitman M, Lysiansky M, Lysyansky P, Friedman Z, Tyomkin V, Fuchs T, et al. Circumferential and Longitudinal Strain in 3 Myocardial Layers in Normal Subjects and in Patients with Regional Left Ventricular Dysfunction. J Am Soc Echocardiogr. 2010;23(1):64–70.
|
| 14. |
Lipiec P, et al. Transthoracic echocardiography in adults – guidelines of the Working Group on Echocardiography of Polish Cardiac Society. Kardiol Pol. 2018; 76(2):488-493.
|
| 15. |
Iio C, Inoue K, Nishimura K, Fujii A, Nagai T, Suzuki J, et al. Characteristics of Left Atrial Deformation Parameters and Their Prognostic Impact in Pa- tients with Pathological Left Ventricular Hypertrophy: Analysis by Speckle Tracking Echocardiography. Echocardiography. 2015;32(12):1821–30.
|
| 16. |
Thorstensen A, Dalen H, Hala P, Kiss G, D’hooge J, Torp H, et al. Three-Dimensional Echocardiography in the Evaluation of Global and Regional Function in Patients with Recent Myocardial Infarction: A Comparison with Magnetic Resonance Imaging. Echocardiography. 2013;30(6):682–92.
|
| 17. |
Fujimoto K, Inoue K, Saito M, Higashi H, Kono T, Uetani T, et al. Incremental value of left atrial active function measured by speckle tracking echocardiography in patients with hypertrophic cardiomyopathy. Echocardiography. 2018;35(8):1138–48.
|
| 18. |
Aly MFA, Brouwer WP, Kleijn SA, van Rossum AC, Kamp O. Three-dimensional speckle tracking echocardiography for the preclinical diagnosis of hypertrophic cardiomyopathy. Int J Cardiovasc Imaging. 2014;30(3):523–33.
|
| 19. |
Hees PS, Fleg JL, Dong S-J, Shapiro EP. MRI and echocardiographic assessment of the diastolic dysfunction of normal aging: altered LV pressure decline or load? Am J Physiol -Heart Circ Physiol. 2004;286(2):H782–8.
|
| 20. |
PiepoliMF,HoesAW,AgewallS,AlbusC,BrotonsC,CatapanoAL,etal.2016EuropeanGuidelinesoncardiovasculardiseasepreventioninclinical practice. Eur Heart J. 2016;37(29):2315–81.
|
| 21. |
KirchhofP,BenussiS,KotechaD,AhlssonA,AtarD,CasadeiB,etal.2016ESCGuidelinesforthemanagementofatrialfibrillationdevelopedin collaboration with EACTS. Eur Heart J. 2016;37(38):2893–962.
|
| 22. |
NaguehSF,SmisethOA,AppletonCP,ByrdBFIII,DokainishH,EdvardsenT,etal.RecommendationsfortheEvaluationofLeftVentricularDias- tolic Function by Echocardiography: An Update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. Eur Heart J – Cardiovasc Imaging . 2016;17(12):1321–60.
|
| 23. |
LangRM,BadanoLP,Mor-AviV,AfilaloJ,ArmstrongA,ErnandeL,etal.RecommendationsforCardiacChamberQuantificationbyEchocardiog- raphy in Adults: An Update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. Eur Heart J – Cardiovasc Imaging. 2015;16(3):233–71.
|
| 24. |
VieiraMJ,TeixeiraR,GonçalvesL,GershBJ.LeftAtrialMechanics:EchocardiographicAssessmentandClinicalImplications.JAmSocEchocardi- ogr. 2014;27(5):463–78.
|
| 25. |
SarvariSI,HaugaaKH,StokkeTM,AnsariHZ,LerenIS,HegbomF,etal.Strainechocardiographicassessmentofleftatrialfunctionpredictsrecur- rence of atrial fibrillation. Eur Heart J – Cardiovasc Imaging. 2015;17(6):660–7.
|
| 26. |
ZhangL.Hypertrophiccardiomyopathy:Canthenoninvasivediagnostictestingidentifyhighriskpatients?WorldJCardiol.2014;6(8):764-770.
|
| 27. |
Voigt J-U, Pedrizzetti G, Lysyansky P, Marwick TH, Houle H, Baumann R, et al. Definitions for a Common Standard for 2D Speckle Tracking Echocardiography: Consensus Document of the EACVI/ASE/Industry Task Force to Standardize Deformation Imaging. J Am Soc Echocardiogr. 2015;28(2):183–93.
|









