Abstract
Background: Anemia is a major public health concern in Sub-Saharan Africa, but data from rural Madagascar are scarce. This study assessed monthly anemia prevalence and demographic and seasonal patterns in northern Madagascar.
Material and methods: We conducted a retrospective cross-sectional analysis of hemoglobin levels of patients routinely tested at a secondary-level rural referral clinic in Manerinerina (Madagascar) between January and December 2024. Hemoglobin was measured using automated methods and anemia in adults was defined as < 12.0 g/dL. Monthly prevalence and demographic associations were examined using chi-square tests and odds ratios (ORs).
Results: Among the 1714 patients, 45.4% (n = 778) were anemic. Children < 15 years of age had the highest prevalence (boys 79.3%, girls 74.6%) and more than six-fold higher odds of anemia than adults aged 15-49 (OR 6.24, 95% CI 4.80-8.11). Women were more often anemic than men (48.9% vs. 40.6%; OR 1.40, 95% CI 1.15-1.70). Monthly prevalence peaked during the rainy season: December (60.2%) to March (24.0%).
Conclusions: Anemia is highly prevalent in this rural Malagasy population, particularly among children and women of reproductive age. Pronounced seasonal variation suggests that cyclical food insecurity substantially increases vulnerability to anemia. Strengthening routine screening and access to qualified healthcare providers should be prioritized, particularly during the rainy season.
Citation
Kasprowicz D, Rajaomalala F. Nearly half of the patients are anemic: seasonal and demographic patterns in rural Madagascar. Eur J Transl Clin Med.Introduction
Anemia is a reduction in red blood cell number or hemoglobin concentration that impairs oxygen transport [1]. According to the World Health Organization (WHO) 2024 guidelines, diagnostic cut-off values vary by age, sex, physiological status and altitude; at sea level, anemia is defined as hemoglobin < 11.0 g/dL in children < 5 years of age and pregnant women, and < 12.0 g/dL in non-pregnant women [2]. This condition remains a major public-health problem in many low- and middle-income countries, particularly in sub-Saharan Africa where nutritional deficiencies, infections and parasitic diseases are highly prevalent [3].
Due to its substantial impact on child development, maternal health and economic productivity, anemia reduction has been designated a global health priority. The WHO aims to decrease anemia in women of reproductive age by 50% by 2030 [4]. Meanwhile, the United Nations incorporated anemia control into its Sustainable Development Goal 2 (target 2.2), which calls for eliminating all forms of malnutrition by 2030 [5]. The African region continues to report the highest anemia prevalence worldwide – 43.0% in 2000, 36.0% in 2012 and 36.5% in 2023 [3]. Despite the fact that the recent WHO estimates indicate that anemia affects over one-third of women aged 15-49 years in Madagascar, national and region-specific data (particularly from the rural northern areas) from that country remain limited [6]. Such region-specific estimates are required to identify periods of increased vulnerability, support resource allocation and contextualize local epidemiological patterns within national trends. Therefore, the aim of this study was to assess monthly anemia prevalence over a full calendar year among patients attending a rural referral clinic in northern Madagascar and to contextualize these findings within national estimates.
Material and methods
This retrospective cross-sectional study analyzed routine clinical and laboratory data collected over a 12-month period (January–December 2024) at Clinique Médicale Beyzym (CMB), a secondary-level referral facility located in Manerinerina (Ambatoboeny District of northwestern Madagascar 16°17′02″ S, 47°17′05″ E). This clinic serves a predominantly rural population within the Boeny Region, characterized by low population density, widespread poverty, agrarian livelihood, and limited access to formal healthcare and diagnostic services.
All hemoglobin measurements recorded as part of routine clinical care during the study period were eligible for inclusion. No additional exclusion criteria were applied. Data were extracted from monthly medical activity reports and laboratory registers.
Blood sampling and hemoglobin assessment
Venous blood was collected into EDTA tubes using a vacuum-assisted blood collection system by trained nursing personnel in accordance with internal Standard Operating Procedures (SOP). Hemoglobin levels were measured using a Mindray M-30 hematology (Mindray Bio-Medical Electronics Co., Shenzhen, China). All laboratory procedures were performed by qualified diagnostic staff, and the final validation of results was conducted by a supervising clinical biologist responsible for quality assurance. Manual May-Grünwald blood smears were prepared as part of routine internal quality control procedures.
Definition and classification of anemia
Although the WHO 2024 guidelines state that hemoglobin thresholds for anemia vary by age, sex, and physiological status, a simplified operational threshold (< 12.0 g/dL) was applied in routine clinical practice at the CMB for all adult patients, regardless of sex [2]. For consistency with local diagnostic practice and to ensure a coherent analytical framework, in this study we adopted the same uniform cut-off.
Statistical analysis
Monthly anemia prevalence was calculated as the proportion of individuals with hemoglobin concentrations < 12.0 g/dL. Differences across months were assessed using the chi-square test (χ²), and effect size was estimated using Cramér’s V. Data entry and organization were performed using Apple Numbers version 6.1 (Apple Inc., Cupertino, California, USA). All statistical analyses were conducted using Python version 3.11 (Python Software Fundation, Wilmington, Delaware, USA) with the SciPy library (scipy.stats).
Ethical considerations
The study protocol was authorized by the Ambatoboeny District Public Health Office and approved by the Hospital Supervisory Committee of the CMB. Only anonymized, routinely collected clinical data were used. No names, addresses, dates of birth, or other identifying information were extracted from the patient records. Data were handled exclusively in aggregated form, in accordance with local ethical guidelines and international standards for the secondary use of clinical information.
Results
A total of 1714 patients were included in this analysis. Hemoglobin concentrations < 12.0 g/dL were documented in 45.4% individuals (n = 778), whereas 54.6% (n = 936) had hemoglobin concentrations ≥ 12.0 g/dL. Among the anemic patients, 57.2% (n = 445) had hemoglobin concentrations between 10.0 and 11.9 g/dL, 28.3% (n = 220) had 8.0 to 9.9 g/dL and 14.5% (n = 113) had hemoglobin < 8.0 g/dL (Table 1).
Table 1. Distribution of hemoglobin concentrations among anemic individuals
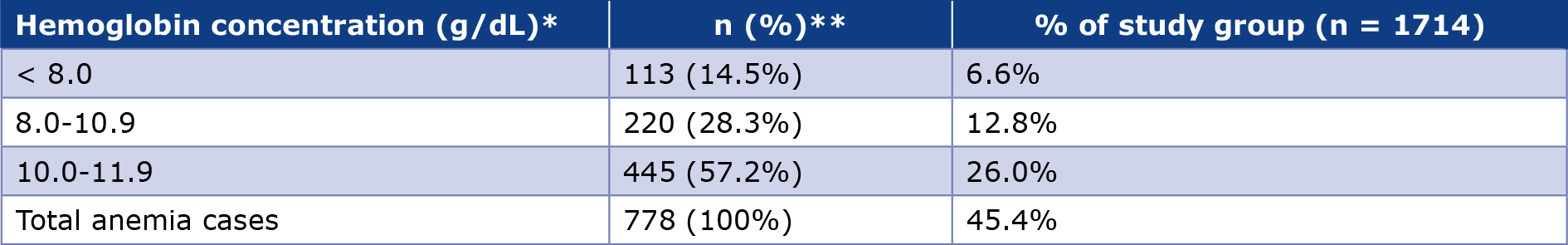
* Anemia was defined according to the WHO hemoglobin guidelines [2].
** Percentages were calculated within the subgroup of individuals with anemia (n = 778).
Anemia was more frequently observed among women than men. Among the 988 women, 48.9% (n = 483) were anemic, compared with 40.6% of the 726 men (n = 295) (χ² = 11.50, df = 1; p = 0.0007). Women had higher odds of anemia than men (OR 1.40, 95% CI 1.15-1.70), although the effect size was small (Cramér’s V = 0.08). Sex-specific hemoglobin distributions are shown in Table 2.
Table 2. Hemoglobin distribution by sex
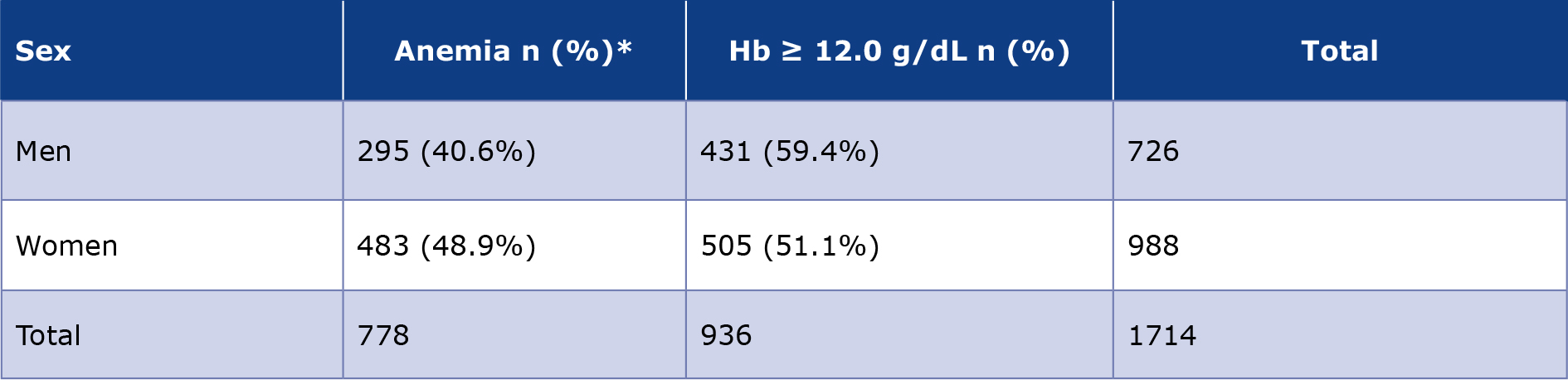
* Anemia was defined as hemoglobin (Hb) < 12 g/dL. Percentages were calculated for each sex (row).
Marked differences were also observed within specific age-sex strata. Among children 5-14 years of age, anemia affected 79.3% of boys and 74.6% of girls. In adults 15-49 years of age, anemia was identified in 20.3% of men and 43.8% of women, demonstrating a substantially higher burden among women of reproductive age. In adults ≥ 50 years, anemia prevalence reached 40.4% among men and 34.8% among women. Overall, age group was strongly associated with anemia (χ² = 218.70, degrees of freedom = 2; p < 0.0001), with a large effect size (Cramér’s V = 0.36). Children had over six-fold higher odds of anemia than adults aged 15-49 years (odds ratio (OR) 6.24, 95% confidence interval (CI) 4.80-8.11). Fully stratified distributions of hemoglobin concentrations, including anemia prevalence with 95% CI, are presented in Table 3.
Table 3. Age- and sex-specific distribution of hemoglobin concentrations with anemia prevalence
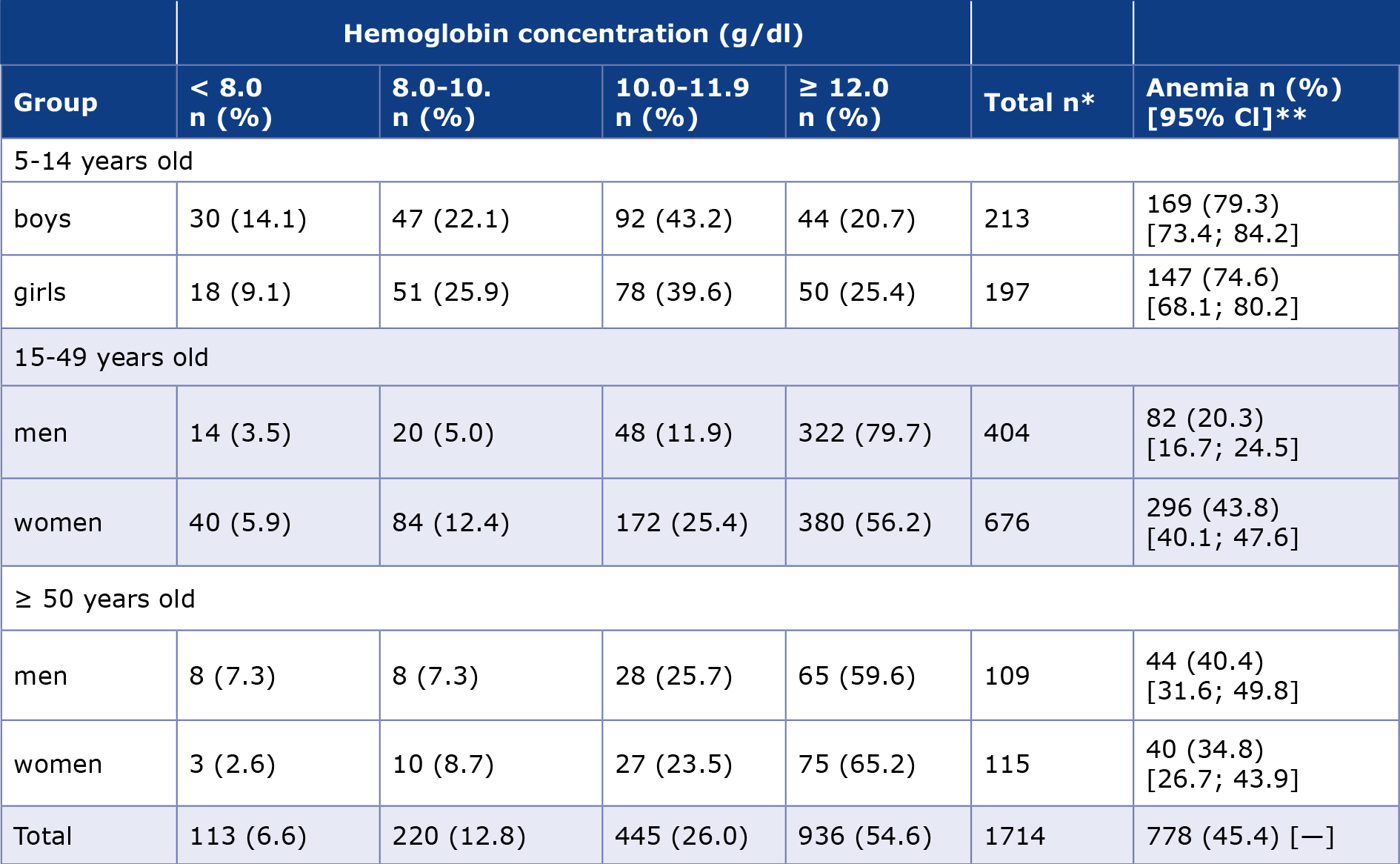
* Percentages within hemoglobin categories are calculated within each age- and sex-specific subgroup (row-based percentages).
** Anemia prevalence is reported as the proportion of individuals with hemoglobin values below the WHO–defined thresholds within each subgroup; 95% confidence intervals were calculated using the binomial method.
Anemia prevalence varied during the year, ranging from 24.0% in March to 60.2% in December. Higher proportions were observed in January (58.5%), February (57.3%), May (57.4%), November (58.0%), and December (60.2%), while lower proportions were recorded in March (24.0%) and July (34.2%). Month of the year was significantly associated with anemia (χ² = 99.02, df = 11; p < 0.0001), with a moderate effect size (Cramér’s V = 0.24). The monthly distribution of hemoglobin categories, expressed as proportions of all patients, is shown in Figure 1.
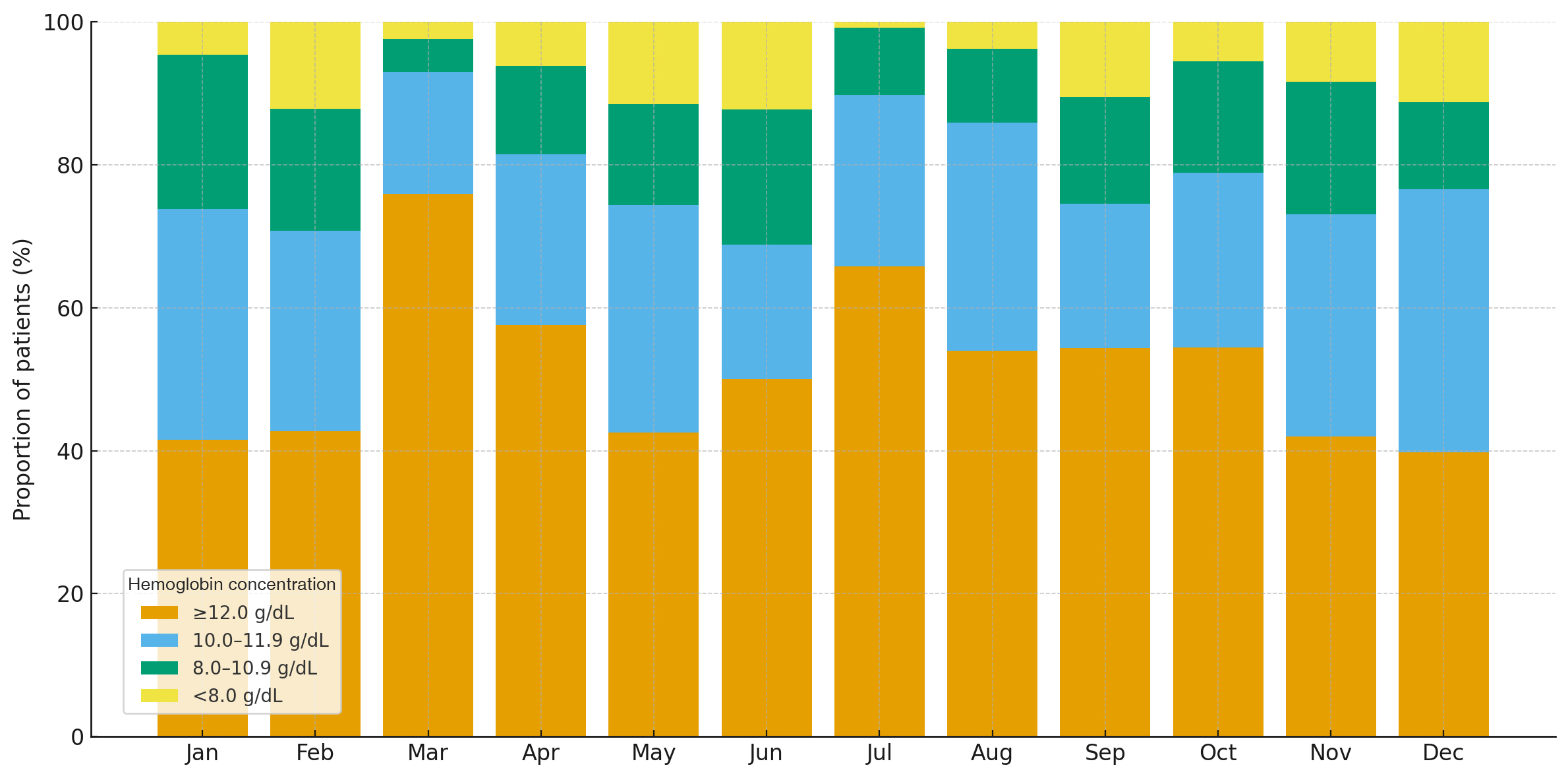
Figure 1. Monthly distribution of hemoglobin concentration expressed as proportions of all patients
Overall, anemia affected nearly half of the studied population, with statistically significant differences across sex and age groups and marked variation across months of the year. The highest burden was observed among children < 15 years of age, followed by older adults (≥ 50 years of age) and women of reproductive age (15-49 years of age). Several monthly anemia proportions exceeded 55% and coincided numerically with periods of increased rainfall, suggesting a potential link between seasonal environmental conditions and anemia burden in this rural population.
Discussion
This study demonstrated a substantial burden of anemia in the rural population of northwestern Madagascar, with nearly half of all patients presenting with hemoglobin concentrations below the WHO-defined thresholds. The prevalence of anemia was particularly high among children < 15 years of age (almost 4 out of 5 were affected), indicating the most vulnerable demographic subgroup. Marked sex-related differences were also evident: women showed a significantly higher prevalence of anemia than men, and this disparity was most pronounced among women of reproductive age. In older adults, anemia remained common in both sexes, affecting more than one-third of individuals. Moreover, although anemia was observed throughout the entire year, its prevalence peaked in December, coinciding with the onset of the rainy season – a period typically associated with reduced food security and limited access to affordable nutritional resources in many rural areas of Madagascar. These findings underscore the heterogeneous distribution of anemia across demographic strata and highlight the relevance of age-sex-specific and seasonal patterns when assessing the local epidemiology of anemia in resource-limited settings.
When placed within the broader sub-Saharan African context, the anemia prevalence identified in our study stands notably above the expectations. Across sub-Saharan Africa, anemia remains one of the most persistent public health challenges, with children and women of reproductive age carrying the greatest burden despite global improvement efforts [7]. Regional analyses consistently show that anemia prevalence among children often exceeds 60%, is substantially higher in rural areas and these patterns are attributed to chronic undernutrition, high rates of infectious diseases, and limited healthcare access [8]. Among women, long-term data reveal only slow and uneven declines in anemia prevalence, with many African countries exhibiting stagnation linked to persistent poverty, food insecurity, and constrained availability of micronutrient supplementation [9]. These structural determinants are further compounded by environmental and seasonal pressures, including fluctuating food supply, rainfall-linked disease transmission patterns, and cyclical nutritional shortages all of which can produce sharp local variations in anemia prevalence not captured by the national or continental averages [10]. In this context, the markedly higher rates detected in our study, together with the clear seasonal peak, suggest that localized socioeconomic and ecological stressors may amplify anemia risk far beyond the trends typically reported for sub-Saharan Africa.
The disproportionately high prevalence of anemia we observed among children aligns with previous findings from Madagascar, where pediatric populations consistently demonstrate substantial hematologic vulnerability. Studies conducted across different regions of the country report anemia in 58-100% of examined children, often with a considerable share of moderate or severe cases [11-14]. For instance, a recent hematological assessment in northern Madagascar found that all participating children were anemic, with nearly 80% classified as moderately anemic and approximately 20% severely anemic [11]. Similar trends were reported in preschool and early school-age populations, where more than half of children had hemoglobin concentrations < 12 g/dL [12], and nearly 60% of children were anemic in larger community-based cohorts [13]. Investigations focusing on younger children (particularly those under 10 years of age) also documented high rates of anemia associated with chronic undernutrition, recurrent infections, and limited dietary diversity, further underscoring the systemic nature of this problem [14]. Importantly, most Malagasy pediatric studies, consistent with our findings, report minimal sex differences and limited associations between anemia and narrow age brackets within childhood, although some analyses show that hemoglobin concentrations tend to be lowest among children < 5 years of age [12]. This relationship between malnutrition and hematologic impairment is consistent with broader analyses of feeding challenges and metabolic vulnerabilities among malnourished children in low-resource settings, including Madagascar [15]. Researchers have demonstrated that although parasitic infections (including schistosomiasis and intestinal helminthiasis) are common, they have an inconsistent relationship with hemoglobin concentrations, thus suggesting that pediatric anemia in Madagascar is driven by multifactorial determinants rather than single etiologies [12, 16]. Collectively, these data confirm that anemia is a pervasive and entrenched health burden among Malagasy children, and its exceptionally high prevalence in our results is fully consistent with national pediatric trends.
The high prevalence of anemia among women of reproductive age observed in our data closely reflects the substantial burden documented across sub-Saharan Africa. Recent WHO estimates for Madagascar show that anemia in women aged 15-49 years has remained persistently high over the past decade, increasing from 32.1% in 2010 to 33.3% in 2015 and 35.3% in 2020, reaching 37.2% in 2023 – one of the highest levels reported globally, with wide uncertainty intervals indicative of pronounced regional disparities [6]. Our 2024 data exceed these national estimates, suggesting that women in remote rural districts may experience considerably greater vulnerability than reflected in national averages. This aligns with evidence from large African meta-analyses, which consistently demonstrate that anemia affects between one-third and one-half of women of reproductive age across the continent, with several analyses estimating pooled prevalences ranging from 38% to over 50% depending on geographic region and socioeconomic context [17-18]. Key determinants identified across African studies include chronic undernutrition, iron deficiency, malaria, intestinal parasitic infections, short interpregnancy intervals, adolescent pregnancies, and limited access to antenatal supplementation programs [19].
More recent analyses further highlight the substantial contribution of food insecurity, poor dietary diversity, and cyclical shortages of staple foods – factors that disproportionately affect rural populations [20]. Environmental stressors, including seasonal peaks in infectious diseases and fluctuations in agricultural productivity, have also been shown to exacerbate anemia risk in African women [21]. Taken together, these continental trends suggest that the disproportionately high prevalence recorded in our study represents an intensified expression of broader African patterns, magnified by the intersection of poverty, environmental instability, and limited health system reach in rural Madagascar.
The marked seasonal variation observed in our dataset, with anemia peaking between November and March, corresponds to the well-documented “lean season” in many rural regions of sub-Saharan Africa. This period, which follows the depletion of stored crops and precedes the next harvest, is characterized by reduced food availability, sharp increases in staple food prices, and widespread dietary insufficiency [22]. In the context of Madagascar, this seasonal window often represents the most acute phase of food insecurity, during which families experience prolonged caloric deficits and markedly limited access to iron-rich or protein-dense foods [23-24]. Such nutritional constraints are strongly reflected in our findings, which show the highest proportions of anemic individuals precisely during months when agricultural production is minimal and household food reserves are exhausted.
The rainy season further compounds these vulnerabilities: although rainfall in the Boeny region is relatively modest compared with other tropical zones, these intermittent wet conditions create favorable breeding sites for the Anopheles mosquitoes, contributing to seasonal surges of malaria [25]. Malaria-associated hemolysis and inflammation, superimposed on chronic undernutrition, may further exacerbate anemia risk during this period [26]. Taken together, these overlapping environmental and socioeconomic pressures suggest that seasonal instability in food security (rather than isolated infectious or nutritional factors) acts as a dominant driver of the pronounced fluctuations in anemia prevalence observed in this rural Malagasy population.
From a public health perspective, the high burden of anemia observed in this population underscores the potential relevance of integrated intervention strategies. Evidence from low-resource settings indicates that effective anemia control requires multifaceted approaches, including iron supplementation, food fortification, maternal and child nutrition programs, and infection control measures, rather than single-component interventions alone [27-29]. Importantly, the effectiveness and safety of these strategies are highly context-dependent. In malaria-endemic and infection-prone regions, indiscriminate iron supplementation may be associated with limited benefit or potential risks, thus emphasizing the need for targeted, screening-based approaches [28, 30]. In such settings, combining nutritional support with infection control, deworming, and strengthened primary healthcare services may offer more sustainable population-level benefits [27, 29]. While our study was not designed to assess the etiology of anemia or intervention efficacy, these findings highlight the need for context-adapted public health strategies and further research to inform evidence-based anemia control programs in rural Madagascar and comparable settings.
Overall, the interplay of nutritional deficits, infection dynamics, and seasonal instability appears to shape the anemia burden observed in this population, pointing to a multifactorial problem that warrants further investigation.
Limitations
Our study has several important limitations. First, we analysed routinely collected clinical data from a single rural referral clinic. As such, our findings reflect anemia patterns among individuals seeking care rather than providing population-level prevalence for the Ambatoboeny District. However, clinic-based datasets remain highly informative for detecting local trends, seasonal variations, high-risk demographic groups, and they offer essential insight in settings where community-level epidemiological data are scarce.
Second, although hemoglobin measurements were performed using standardized procedures and subjected to internal quality control, we relied on a simplified clinical threshold for anemia classification that does not fully align with ageand sex-specific WHO cut-offs. This operational choice may have led to some misclassification of anemia status, particularly among men and younger adolescents, and could have influenced comparisons with external datasets. Furthermore, the analysis was based on single time-point hemoglobin assessments, without complementary biomarkers such as the serum concentrations of ferritin, C-reactive protein, vitamin B12, folate or transferrin saturation. Consequently, we were unable to distinguish iron-deficiency anemia from anemia of inflammation, hemoglobinopathies, or other etiologies.
Third, because we did not aim to determine the etiology of anemia, routinely collected registers did not include information about nutritional biomarkers and co-morbidities (e.g. malaria, helminthic infections, protozoal diseases). While these factors are known to contribute to anemia in Madagascar (as shown in previous studies from the region), our analysis was designed to describe overall anemia prevalence and its demographic/seasonal patterns rather than to attribute causality. Although the absence of etiological variables limits the interpretation of mechanisms, it does not affect our primary objective of quantifying anemia distribution in this population. Despite these limitations, our study provides much-needed, context-specific evidence from a specific region of Madagascar, where health data are limited. The observed age-, sex- and season-related patterns of anemia prevalence offer a valuable basis for future community-based or longitudinal investigations.
Conclusions
This study confirms that anemia is a major and persistent health problem in rural Madagascar, with a particularly high burden among children < 15 years of age and women of reproductive age. The observed seasonal variation in hemoglobin levels suggests that periods of increased vulnerability, potentially related to food insecurity and the burden of infectious and parasitic diseases, are associated with lower hemoglobin concentrations. These findings underscore the need to strengthen routine hemoglobin screening in primary care, particularly during periods when population-level vulnerability is expected to peak.
Given that anemia prevalence in this rural setting exceeds national and regional estimates, targeted public health interventions are urgently needed. Priorities should include improving access to qualified healthcare providers (e.g. who are capable of evaluating anemia, identifying its likely contributors, and initiating appropriate management), as well as expanding local monitoring systems to better capture anemia trends in remote communities. Further research is essential to clarify the multifactorial drivers of anemia in these settings and to inform the development of context-appropriate strategies for prevention, early detection, and clinical decision-making.
Acknowledgements
The authors express their gratitude to all the clinical and laboratory staff of Clinique Médicale Beyzym in Manerinerina for their daily commitment to patient care and their invaluable assistance in maintaining high-quality medical records. In particular, we wish to thank Razanatsimialintsoa Fidelia for her meticulous work in organizing and validating monthly laboratory reports, Ravolanjafy Razely for her support in retrieving and interpreting ward activity registers, and Dr Mazava Sabbat Dieudonné (head physician) for his continuous clinical guidance and supervision of diagnostic decision-making.
We are also grateful to the District Public Health Office of Ambatoboeny staff for their administrative support and for authorizing the use of routine clinical data for this analysis. Finally, we sincerely thank all the patients and their families who sought care at Clinique Médicale Beyzym; without their trust in this clinic, our study would not have been possible.
Funding
The authors did not receive any specific external funding for this study.
Conflicts of interest
None to report.










